Trethera Announces Appointment of R. Boyd Quinnell to Board of Directors
Los Angeles, January 18, 2023 — Trethera Corporation (“Trethera”), a clinical-stage biopharmaceutical company committed to developing novel drugs targeting nucleotide metabolism for the treatment of cancer and autoimmune diseases, announced today the appointment of Boyd Quinnell, CPA, MBA, to the Trethera Board of Directors as an independent director. A veteran healthcare executive with over 30 years of experience in executive and financial leadership, Mr. Quinnell will also chair the Board’s Audit Committee.
“We are pleased to welcome Boyd to the Trethera Board,” said Dr. Ken Schultz, Chairman and Chief Executive Officer of Trethera. “Boyd’s deep financial expertise in medical technology global businesses with strong regulatory oversight will be invaluable to Trethera as we continue to advance towards first product revenue. His experience commercializing and scaling innovative technologies to improve health outcomes aligns well with our mission, making him an ideal board addition.”
“I am delighted to join the Board at this exciting time in Trethera’s growth,” said Mr. Quinnell. “With promising Phase 1 clinical data in oncology and a strong potential to treat a wide array of autoimmune diseases, Trethera is well positioned to execute its mission and clinically advance its lead drug candidate.”
Mr. Quinnell, 61, is the former Chief Financial Officer and General Manager for the US division of Biotronik, a medical device company generating over $400M in annual revenue, where he served from 2015 through 2019. Prior to Biotronik, Mr. Quinnell worked for 12 years at increasing levels of responsibility at the world’s largest medical device company, Medtronic, exiting as Controller and Senior Director of Finance. He began his career as a senior consultant at KPMG, one of the Big Four accounting firms. Mr. Quinnell received a Bachelor of Arts (BA) degree in business from the University of Hawaii and a Master of Business Administration (MBA) degree from Cornell University. He holds an active certified public accountant (CPA) license. He has served on non-profit boards for the American Heart Association and the charity Food on Foot. Mr. Quinnell remains active in the Los Angeles community in his efforts to alleviate homelessness.
About Trethera
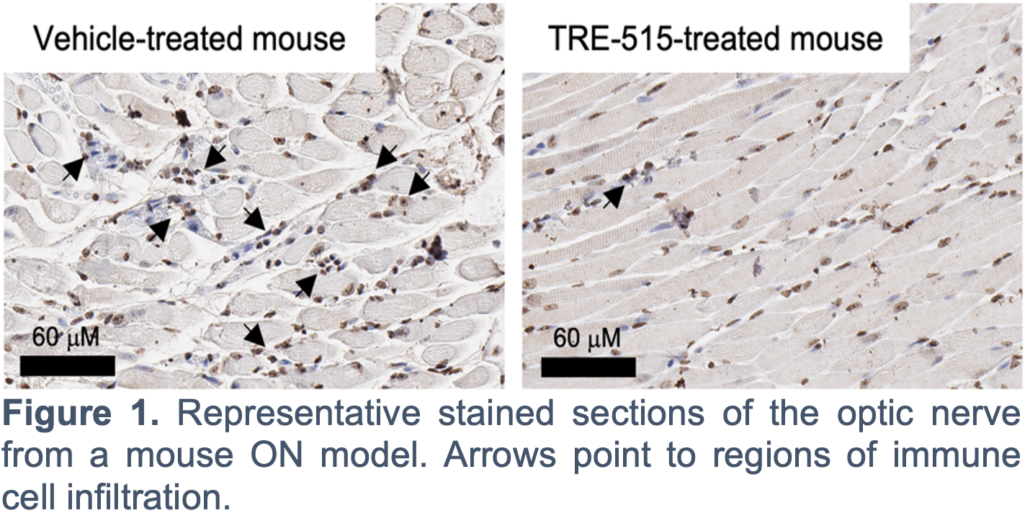
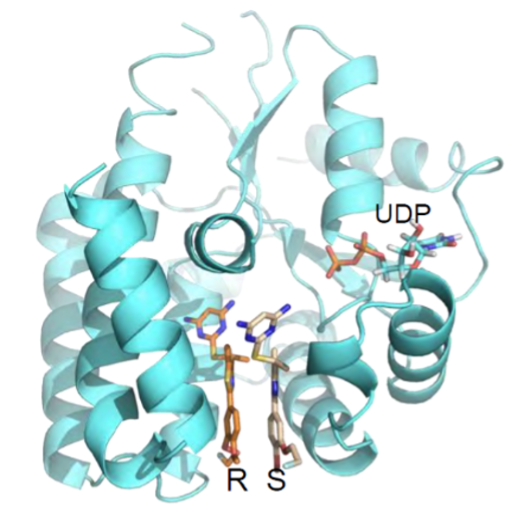
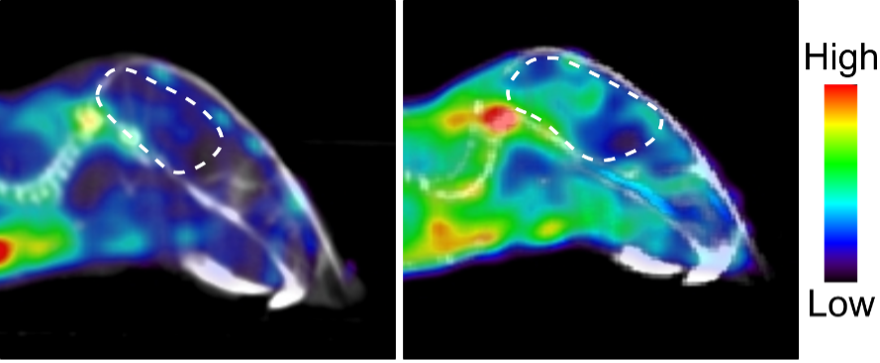
Trethera is a clinical stage privately held biopharmaceutical company dedicated to pioneering the development of novel treatments for autoimmune diseases and cancers. Founded by prominent UCLA scientists, Trethera is led by experienced management and board members. Trethera’s innovative approach to targeting nucleotide metabolism led to the development of TRE-515, an orally administered capsule twice designated by the FDA as an Orphan Drug. TRE-515 is a first-in-class clinical stage drug that inhibits deoxycytidine kinase (dCK), the rate-limiting enzyme in the nucleoside salvage pathway, one of two biosynthetic pathways that generate DNA precursors. It is believed that some forms of cancer may be preferentially dependent on the salvage pathway to support tumor growth, and certain autoimmune diseases, such as multiple sclerosis, might also respond to TRE-515 treatment. Trethera is developing TRE-515 for use as a monotherapy or in combination, to precisely target a metabolic vulnerability of cancer or autoimmune diseases that will transform outcomes for patients.
For more information, please visit us at trethera.com or e-mail Investor Relations at ir@trethera.com.
Note on Forward-Looking Statements
All statements other than statements of historical facts included in this press release that address activities, events or developments that Trethera believes or anticipates will or may occur in the future are “forward-looking statements,” which may often, but not always, be identified by the use of such words as “may,” “might,” “will,” “will likely result,” “would,” “should,” “estimate,” “plan,” “project,” “forecast,” “intend,” “expect,” “anticipate,” “believe,” “seek,” “continue,” “target” or the negative of such terms or other similar expressions. Although Trethera has a reasonable basis for the forward-looking statements contained herein, Trethera cautions that such statements are based on current expectations about future events and are subject to risks, uncertainties and factors relating to medical and scientific research, all of which are difficult to predict and many of which are beyond Trethera’s control, that may cause actual results to differ materially from those expressed or implied by the forward-looking statements in this press release. These potential risks and uncertainties include, without limitation: the extent to which development of any novel cancer therapies or therapies for autoimmune diseases succeeds; whether Trethera would obtain the necessary regulatory approvals to commence human trials or commercialize TRE-515 or any novel therapies resulting from such research; Trethera successfully implementing its growth strategy, including that relating to its disease therapies; the effects of the global Covid-19 pandemic; changes in economic conditions; competition; and risks and uncertainties applicable to the business of Trethera. The statements in this press release speak only as of the date hereof and Trethera does not undertake any obligation to update, amend or clarify these forward-looking statements whether as a result of new information, future events or otherwise. The Company intends that all forward-looking statements be subject to the safe-harbor provisions of the Private Securities Litigation Reform Act of 1995.