Trethera Receives $1.6 Million National Eye Institute Grant for Continued Advancement of TRE-515 Optic Neuritis Program
Los Angeles, October 19, 2022 — Trethera Corporation (“Trethera”), a clinical stage biopharmaceutical company committed to developing novel drugs targeting nucleotide metabolism for the treatment of cancer and autoimmune diseases, announced today that the National Eye Institute (NEI), a division of the National Institutes of Health (NIH), has awarded Trethera a $1.6 million Fast-Track STTR commercialization grant to further develop TRE-515 as a treatment for optic neuritis (ON). TRE-515 is a novel first-in-class drug, which has been shown to prevent demyelination, and even facilitate remyelination, in multiple demyelinating disease mouse models. Furthermore, the FDA has designated TRE-515 an Orphan Drug in the treatment of ON. The grant monies and research activities will be shared in collaboration with scientists at the University of California Los Angeles (UCLA) with support from researchers at Stanford and Harvard.
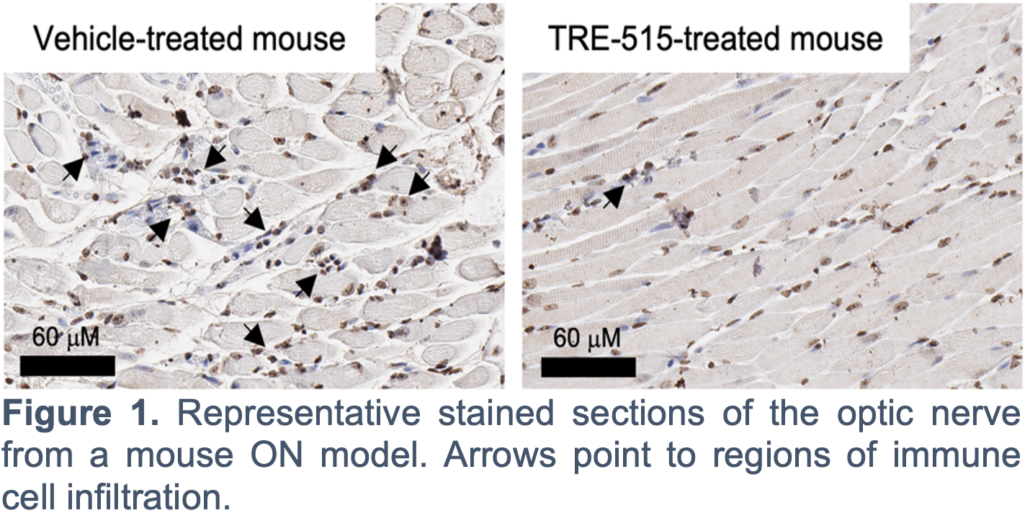
TRE-515 preclinical development activities required for an investigational new drug (IND) application as well as dose response studies in mice. While initial TRE-515 development efforts focused on solid tumors, which has an enrolling Phase 1 clinical trial, preclinical in vivo data along with a growing body of literature supports potential application of TRE-515 in several autoimmune diseases including ON.
“We are excited and grateful for this NEI funding as it allows Trethera to build upon our promising preclinical work conducted to date. New ON treatments fulfill a critical unmet medical need and could benefit the over 100,000 patients in the US who suffer from the disease every year. The last time the FDA approved an ON therapy was over a half century ago,” commented Dr. Ken Schultz, Trethera’s CEO and grant principal investigator. In addition to the NEI grant, Trethera has also raised over $3 million earlier this year through interim financing involving both existing and new investors.
“The use of animal models to evaluate safety and efficacy is essential to the responsible and successful development of novel therapies,” said Dr. Peter M. Clark, Associate Professor in charge of the UCLA-based research. “The preclinical data generated to date in our ON program is very encouraging, and we believe that the work supported by this new grant will provide important information that will help to optimize the design of human clinical trials for ON.”
ON is an autoimmune disease caused by inflammatory demyelination of the optic nerve, with the typical patient being a woman in her early 30s. ON patients experience acute, unilateral, painful vision loss. Cases of ON have a close association with multiple sclerosis (MS). Although steroids accelerate the initial recovery rate of ON, they have no effect on long-term visual outcomes or whether ON patients develop MS. ON has been also associated with neuromyelitis optica and systemic disorders such as lupus.
“For some patients, ON can be self-resolving, but for others ON can lead to lifelong disability. In all ON cases, the threat of future conversion to MS remains. TRE-515 could potentially provide significant benefit to ON patients beyond the available therapeutic options, especially those taking long-term steroids. Any drug that could improve these outcomes for patients would be game changing,” commented Trethera Scientific Advisory Board member and grant supporter, Dr. Larry Steinman. Dr. Steinman is a noted immunologist and neurologist; a Professor of Neurology, Pediatrics, and Genetics at Stanford University; and a member of both National Academies of Sciences and Medicine.
A discussion summary from the panel of NIH experts that reviewed Trethera’s proposal stated, “Reviewers were highly enthusiastic … TRE-515 holds FDA Orphan Drug Designation for the treatment of ON, which allows for an accelerated timeline … TRE-515 treatments and dCK knockout are not associated with significant toxicities … a high chance of success, considering the preliminary proof-of-concept data and the parallel Phase 1 clinical development of the same molecule for a different indication.”
“A grant award in the Fast Track category is perhaps the most competitive at the NEI, and a mark of distinction,” stated Dr. Michael Levy, Research Director of the Division of Neuroimmunology & Neuroinfectious Disease at Massachusetts General Hospital and Trethera grant supporter. “As an ON treatment has not been approved by the FDA since 1952, I look forward to helping TRE-515 continue its progress to the clinic for this disease.” Dr. Levy is a distinguished ON specialist, an Associate Professor at Harvard Medical School, and a Fellow of the American Academy of Neurology.
TRE-515 is an orally delivered therapeutic engineered to inhibit dCK, the key enzyme in the nucleoside salvage pathway. A common characteristic of tumor cells in solid malignancies and pathological immune cells in autoimmune diseases is that they require elevated nucleotide levels to support accelerated cell division and abnormal activation. Given the rate limiting role of the enzyme dCK in the nucleoside salvage pathway, it is a key target in both malignant cell proliferation and autoimmune lymphocyte activation. In contrast, dCK activity is not required in most healthy adult human cells, suggesting dCK is a highly specific therapeutic target.
About Trethera
Trethera is a clinical stage privately held biopharmaceutical company dedicated to pioneering the development of novel treatments for autoimmune diseases and cancers. Founded by prominent UCLA scientists, Trethera is led by experienced management and board members. Trethera’s innovative approach to targeting nucleotide metabolism led to the development of TRE-515, an orally taken capsule twice designated by the FDA as an Orphan Drug. TRE-515 is a first-in-class clinical stage drug that inhibits deoxycytidine kinase (dCK), the rate-limiting enzyme in the nucleoside salvage pathway, one of two biosynthetic pathways that generate DNA precursors. It is believed that some forms of cancer may be preferentially dependent on the salvage pathway to support tumor growth, and certain autoimmune diseases, such as multiple sclerosis, might also respond to TRE-515 treatment. Trethera is developing TRE-515 for use as a monotherapy or in combination, to precisely target a metabolic vulnerability of cancer or autoimmune diseases that will transform outcomes for patients.
For more information, please visit us at trethera.com or e-mail Investor Relations at ir@trethera.com.
Note on Forward-Looking Statements
All statements other than statements of historical facts included in this press release that address activities, events or developments that Trethera believes or anticipates will or may occur in the future are “forward-looking statements,” which may often, but not always, be identified by the use of such words as “may,” “might,” “will,” “will likely result,” “would,” “should,” “estimate,” “plan,” “project,” “forecast,” “intend,” “expect,” “anticipate,” “believe,” “seek,” “continue,” “target” or the negative of such terms or other similar expressions. Although Trethera has a reasonable basis for the forward-looking statements contained herein, Trethera cautions that such statements are based on current expectations about future events and are subject to risks, uncertainties and factors relating to medical and scientific research, all of which are difficult to predict and many of which are beyond Trethera’s control, that may cause actual results to differ materially from those expressed or implied by the forward-looking statements in this press release. These potential risks and uncertainties include, without limitation: the extent to which development of any novel cancer therapies or therapies for autoimmune diseases succeeds; whether Trethera would obtain the necessary regulatory approvals to commence human trials or commercialize TRE-515 or any novel therapies resulting from such research; Trethera successfully implementing its growth strategy, including that relating to its disease therapies; the effects of the global Covid-19 pandemic; changes in economic conditions; competition; and risks and uncertainties applicable to the business of Trethera. The statements in this press release speak only as of the date hereof and Trethera does not undertake any obligation to update, amend or clarify these forward-looking statements whether as a result of new information, future events or otherwise. The Company intends that all forward-looking statements be subject to the safe-harbor provisions of the Private Securities Litigation Reform Act of 1995.
